Neuroscientists usually investigate one brain at a time. They observe how neurons fire as a person reads certain words, for example, or plays a video game. As social animals, however, those same scientists do much of their work together—brainstorming hypotheses, puzzling over problems and fine-tuning experimental designs. Increasingly, researchers are bringing that reality into how they study brains.
Collective neuroscience, as some practitioners call it, is a rapidly growing field of research. An early, consistent finding is that when people converse or share an experience, their brain waves synchronize. Neurons in corresponding locations of the different brains fire at the same time, creating matching patterns, like dancers moving together. Auditory and visual areas respond to shape, sound and movement in similar ways, whereas higher-order brain areas seem to behave similarly during more challenging tasks such as making meaning out of something seen or heard. The experience of “being on the same wavelength” as another person is real, and it is visible in the activity of the brain.
Such work is beginning to reveal new levels of richness and complexity in sociability. In classrooms where students are engaged with the teacher, for example, their patterns of brain processing begin to align with that teacher’s—and greater alignment may mean better learning. Neural waves in certain brain regions of people listening to a musical performance match those of the performer—the greater the synchrony, the greater the enjoyment. Couples exhibit higher degrees of brain synchrony than nonromantic pairs, as do close friends compared with more distant acquaintances.
But how does synchrony happen? Much about the phenomenon remains mysterious—even scientists occasionally use the word “magic” when talking about it. One straightforward explanation could be that coherence between brains is a result of shared experience or simply a sign that we are hearing or seeing the same thing as someone else. But the newest research suggests that synchrony is more than that—or can be. Only by looking into the brains of all individuals involved in an interaction, says neuroscientist Weizhe Hong of the University of California, Los Angeles, can we start to “fully understand what is going on.”
Researchers are discovering synchrony in humans and other species, and they are mapping its choreography—its rhythm, timing and undulations—to better understand what benefits it may give us. They are finding evidence that interbrain synchrony prepares people for interaction and beginning to understand it as a marker of relationships. Given that synchronized experiences are often enjoyable, researchers suspect this phenomenon is beneficial: it helps us interact and may have facilitated the evolution of sociality. This new kind of brain research might also illuminate why we don’t always “click” with someone or why social isolation is so harmful to physical and mental health.
Resonance
Those tantalizing prospects are why, last December, I put on a pair of hospital scrubs and lay in the tube of a functional magnetic resonance imaging (fMRI) machine at Harvard University. As instructed, I tried to keep as still as possible with my head in a cradle and my left thumb poised on an emergency call button. It was as uncomfortable as I had been warned it would be.
“Are you okay?” asked a muffled voice from the control room next door.
“I’m good,” I lied.
Then a new, louder voice sounded in my earbuds: “Can you hear me?”
This was Sid. He was going to be my conversation partner for the next hour.
We introduced ourselves. I said I was a science journalist. He said he worked in a social neuroscience laboratory at Dartmouth College. Sid and I were communicating via the Internet as we lay in separate brain-imaging machines 130 miles apart.
Instructions flashed on the screens above each of us. Our task was to tell a story together in alternating turns of 30 seconds each. I was to go first using this prompt: “A group of children encounters aliens.”
I launched into a story about children on a school field trip who went for a walk in a park with their teachers and stumbled on the dramatic landing—loud noise, bright lights—of an alien spaceship. Sid had some of the braver children venture closer, led by a boy named Kevin. I added a girl named Annabel who reached out a finger to touch one of the creatures. Sid threw in some hints of ancient connections between the two worlds.
Eventually the counter on the monitor above me flashed: 4 … 3 … 2 … 1 … time was up. New instructions appeared. Now we each had to build our own story in 30-second increments. Between our own increments, we were to listen to the other person’s evolving tale. When that was done, we both had to retell all three stories: our joint creation and the ones we invented separately.
The story Sid and I told together wasn’t terribly original. My solo effort, about a kid who got in trouble, was even less so. But one thing stood out: I found it far more fun to work together than alone—so much so I forgot about my discomfort. When I met Sid in person the next day at Dartmouth, he agreed. He, too, had enjoyed telling a story with me more than telling his own tale.
That seemed fitting to Dartmouth neuroscientist Thalia Wheatley, who had enlisted us in this pioneering study. While Sid and I did our thing, Wheatley, her postdoctoral researcher JD Knotts and Adam Boncz of the Research Center for Natural Sciences in Budapest listened and watched from control rooms at Harvard and Dartmouth while multiple computers recorded what Sid and I said, when we said it and what our brains were doing at the time. The fMRI machines we were in tracked changes in blood flow throughout the brain, which correlate tightly with changes in neural activity. The results of such imaging highlight, albeit indirectly, where in the brain things are happening. For instance, the auditory cortex should be active while a person is listening, but so should areas in the temporal lobe that process language and meaning.
Later the research team would pore over the voluminous data generated, hoping to see the ways two brains, together, change as they interact and might even make something new. “When we’re talking to each other, we kind of create a single überbrain that isn’t reducible to the sum of its parts,” Wheatley says. “Like oxygen and hydrogen combine to make water, it creates something special that isn’t reducible to oxygen and hydrogen independently.”
At least that is the idea. To see whether they can pinpoint that “something special,” the researchers will compare the activity in my and Sid’s brains, and the brains of all the other pairs in the study, second by second, voxel by voxel over the course of our storytelling session, looking for signs of coherence. They will also consider the questionnaires and reports about the experience we and other participants filled out after we emerged from the machines (using questions such as “How much did you like the story you created with your partner?”). Such studies take time, but in a year or so, if all goes according to plan, they will publish their first results.
The initial “hyperscanning” study—two people, two fMRIs—took place at the Baylor College of Medicine in Houston. Neuroscientist Read Montague, now at Virginia Polytechnic Institute and State University, put two people in separate fMRI scanners and recorded their brain activity as they engaged in a simple competitive game. The relatively limited goals of that experiment were to demonstrate the feasibility of following simultaneous activity in two brains and to identify technical hurdles. The results were published in 2002. Since then, the field has gotten better at hyperscanning with fMRI and expanded to other kinds of technology.
Like fMRI, functional near-infrared spectroscopy (fNIRS) tracks changes in oxygen levels in blood flow; because oxygenation increases with energy demands, scientists can use the method to track brain activity. Employing just a cap of lights and sensors—oxygen-rich blood interacts with light differently than less oxygenated blood does—fNIRS is cheaper and less demanding to administer than fMRI. It is, however, also more limited because it reaches only the upper levels of the brain.
Electroencephalography (EEG), another type of scan, zeroes in on timing, recording the speed and sequence of brain activity—focusing on the when more than the where revealed by fMRI. EEG also reflects the relative pace of different types of brain waves or oscillations. Like waves in water, waves in the brain rise and fall in cycles fast and slow. The five common brain-wave types, named alpha, beta, gamma, delta and theta depending on their oscillation rate, signify different states of the brain. At 0.5 to four hertz (one hertz is a full oscillation per second), delta waves usually represent deep, restful sleep. Other waves are fast and choppy—awake and conscious activity is typically associated with beta (13 to 30 Hz) and gamma waves (roughly 30 to 100 Hz).
New studies similar to Wheatley’s aim to go beyond the early findings and ask, for example, whether storytelling pairs who build better stories show more tightly coupled brain activity than those whose efforts fall a little flat. For the findings to count as “extra” during the joint storytelling condition, correlations between brains “should not be linked simply to people speaking or listening and understanding each other on a linguistic level,” says Boncz, who is a co-lead on the study I took part in. “It should be something more.”
To establish the neural underpinnings of interacting brains, neuroscientists are also turning to other species in which they can investigate at deeper levels of neurobiological detail than in humans. Among the social mammals they are studying, some of the most intriguing—and surprising—are squabbling, snuggling, swooping bats.
Bat-brained
It is not hard to find Michael Yartsev’s lab at the University of California, Berkeley. Small, black, plastic bat wings are pinned to the wall by his nameplate as if they were fluttering around his door. Here it is always Halloween. And it was here, in 2019, that Yartsev and postdoctoral researcher Wujie Zhang were the first to show that bat brains synchronize just as human brains do. Although scientists have long studied collective behavior in animals from insects to mammals, they had never reached the level of the brain in this way.
Yartsev’s groundbreaking study showed what is probably the simplest of the multiple levels of meaning synchrony carries: it is a strong signal of social interaction. In bats, it is present only when they are together.
The bats live downstairs, in what Yartsev, who is both a neuroscientist and an engineer, affectionately calls the “bat cave.” He houses around 300 fruit bats in two colonies, one for males, the other females. The walls of the colony rooms are black, and in each there are mesh panels attached to the ceiling and netting spread throughout the room. Upside-down fruit kebabs of cantaloupe and apple hang from the ceiling, as do blue plastic structures for the bats to play in.
Yartsev was drawn to the study of fruit bats because of their vocal learning and communication skills, but he quickly realized they offered a window into sociality, too. Standing in the doorway of a colony room and watching the bats hang out together, it’s not hard to see why. Although they have plenty of room to spread out, the brown-gray mammals, each six to eight inches long, usually huddle in clusters, clinging to the netting or hanging from the mesh.
In the wild, these highly social fruit bats spend their nights foraging for food and much of the day sleeping in big, crowded colonies in caves or trees—sometimes with hundreds or thousands of other bats. While packed in tightly, they squabble over food, sleeping space and mating attempts.
Down the hall from the colony rooms at Berkeley, there’s a large “flight room” for experiments. While Yartsev and I watch, graduate students carry in two plastic containers with lids and release a group of bats. From the control room next door, the animals show up as dots on the computer monitors, looking like remote-control Ping-Pong balls zinging around the room and occasionally coming to rest in odd corners.
Studying free-flying bats as Yartsev does is an exercise in technical precision. Because the bats spend so much time huddled together and fly so quickly, it can be hard to identify them or figure out which bat vocalized. To track location, behavior and brain activity, the scientists outfitted the flight room with 16 cameras and multiple antennas hidden in small white boxes. Tiny transponders hanging around each bat’s neck have microphones that help the team detect which bat is vocalizing, and the cameras detect their locations at resolutions of a centimeter or less. Brain activity is monitored separately via electrodes recording from a variety of brain regions and feeding neural data into tiny, lightweight loggers attached to each bat’s head. When the experiment is done, the information from each logger is uploaded and analyzed.
In Yartsev and Zhang’s 2019 synchrony experiment, they used wireless electrophysiology and other technology to track bats’ behavior and brain activity for about 100 minutes at a time. They saw that the bats’ behavior was roughly correlated—they tended to rest at the same time and be active at the same time. Their active periods included social and nonsocial behaviors such as fighting or grooming themselves or one another.
To compare brain activity, the scientists analyzed a spectrogram of all brain-wave activity. What stood out in the bats was that high-frequency bands (from 30 to 150 Hz) had more power, or prominence, during periods of active behavior, and low-frequency bands (1 to 29 Hz) had more power during rest. It was also immediately obvious—strikingly so—that there were very high levels of interbrain synchrony among the bats, especially at high frequencies. The patterns were so similar that the researchers initially didn’t believe what they were seeing, but the data convinced them. “Here’s signal number one, and here’s signal number two,” Yartsev says. “Just do the correlation between them. It was so incredibly robust, which was very reassuring because it suggested we were looking at something real. We would see it every single time when they were socially interacting.”
When Yartsev and Zhang repeated the experiment by letting the bats fly freely in identical separate chambers rather than in the same social environment, the correlations fell apart. There was no synchrony in the bats’ brain activity, even when the researchers piped in the sound of other bats calling. And there were more intriguing details. In social situations, the correlations increased as bats interacted more. And increases in correlation between brains preceded increases in social interaction—a reflection of the fact that each interaction is a series of decisions, suggesting that brain correlation facilitates interaction.
Yartsev and Zhang concluded that there is something special about social interaction. Synchrony may be a sign of shared cognitive processing, which is the chemical and electrical signaling in the brain that allows individuals to comprehend their environment, communicate and learn.
Neuron by Neuron
Looking at synchrony between bands of brain waves is one way of understanding what’s going on between interacting brains. Another is to look at the activity of specific neurons. “Ultimately our brains are not a soup of averages. They consist of individual neurons that do different things, and they may do opposite things,” U.C.L.A.’s Hong says. Hong and his colleagues were among the first to go looking for this level of detail and study interacting brains neuron by neuron. What they found revealed even more complexity.
Like Yartsev, Hong first doubted that the interbrain synchrony he and his team observed in animals—in their case, mice—was real. He hadn’t yet read the literature on synchrony in humans and told Lyle Kingsbury—at the time a student of Hong’s and the lead scientist on the research and now a postdoctoral fellow at Harvard University—that there must be something wrong. There wasn’t. Using a technology called microendoscopic calcium imaging, which measures changes in induced fluorescence in individual neurons, they looked at hundreds of neurons at the same time. In pairs of interacting mice, they established that synchrony appeared during an ongoing social interaction. Further, synchrony in mouse brains arose from separate populations of cells in the prefrontal cortex, which Hong calls “self cells” and “other cells.” The former encodes one’s own behavior, the latter the behavior of another individual. “The sum of activity of both self and other cells is similar to or correlated with the sum of activity in the other brain,” Hong says.
What they are seeing goes well beyond previous research on so-called mirror neurons, which represent both the self and another. (When I watch you throw a ball, it activates a set of mirror neurons in my brain that would also be activated if I were doing the same thing myself.) In contrast, the self and other cells Hong and Kingsbury discovered encode only the behavior of one individual or the other. All three kinds of cells—mirror, self and other—were present and aligning in the mouse brains.
The mouse study suggested another level of meaning for synchrony: it predicts the outcomes of future interactions. Like bats, mice enjoy the company of other mice and sleep huddled together, but they are a hierarchical species, with some animals more dominant than others. To take advantage of that, Hong and Kingsbury used a standard experiment called a tube test that is much like watching two football teams try to reach each other’s end zones. The researchers placed two animals in a tube, one at each end, and watched them advance toward each other. They wanted to see which mouse gained the most ground on its opponent. The one who got farther was deemed dominant.
Surprisingly, there were higher levels of synchrony between mice who were further apart in social status—one dominant and one submissive—and lower levels between mice closer in rank. (Researchers in China found something similar in human leaders and followers. In a 2015 study, neural synchronization was higher between leaders and followers than between followers and followers.) Once they recognized the role of social status in their experiment, Hong and Kingsbury could use the levels of synchrony they observed to predict within a few minutes of a 15-minute interaction whether one mouse would dominate and how much more progress it would make.
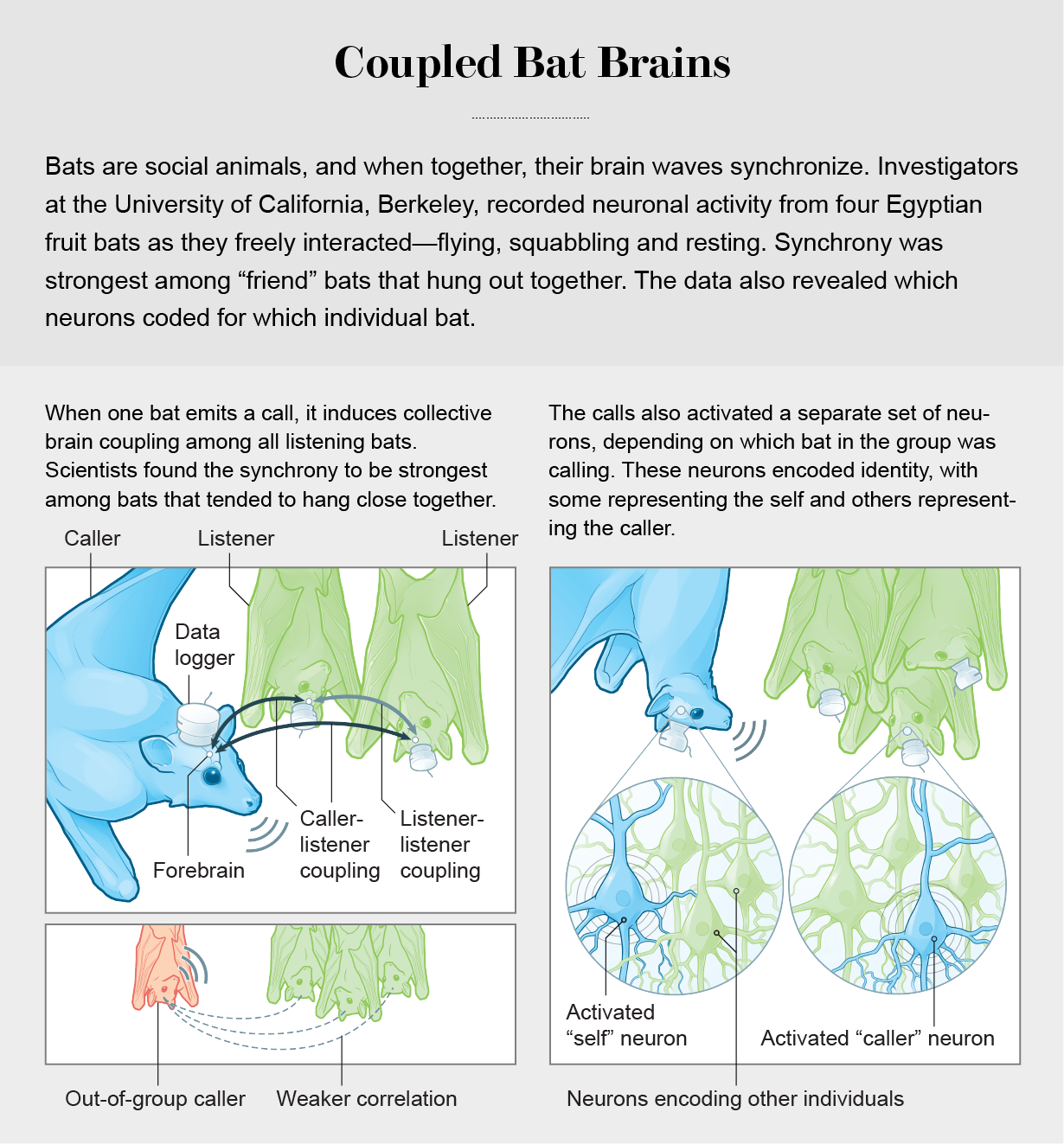
It’s not entirely clear how hierarchical bats are, but they do have preferred companions. Yartsev and his team noticed that most of their bats tended to cluster together, but there were a few that spent their time a little off to the side. The researchers set out to see whether there were differences in levels of correlation when “in-cluster” and “out-of-cluster” bats vocalized. This time, in addition to recording brain activity at the level of frequency bands, they also recorded the activity of individual neurons in the brains of four bats simultaneously as they flew in groups of four, five and eight. A 2021 study led by Maimon Rose and Boaz Styr, then both members of Yartsev’s lab, revealed that when one bat emits a call, it induces collective brain coupling among all listening bats. And as in the mice, separate sets of neurons became active depending on which bat in the group vocalized, meaning individual neurons in the bats’ brains encoded identity, with some representing the self and others representing other individuals. The signals were so distinct that the scientists could tell which bat was calling just by looking at the recordings of neural activity. Correlation among brains was visible in all the bats, but it was strongest when calls came from “friendlier” bats—those that clustered together more often.
The bat and mouse studies were technically very different, but “the two stories are surprisingly similar,” Hong says. “This is the exciting part of science when you see someone else’s work support the conclusions we have [made] independently.”
Beyond Synchrony
The goal of the latest human studies, such as the one Wheatley invited me to join, is not just to explore synchrony more deeply but to go beyond it. Wheatley, who with four other Dartmouth scientists is establishing the college’s Consortium for Interacting Minds, believes that asking when we are in sync with someone else is “a pretty limited way to think about two minds coming together.” More interesting, Boncz says, would be to see whether brains can align at the level of understanding. “We think there could be synchrony, for example, when people understand perhaps even different stimuli the same way, if they have some sort of higher-level meaning that they share.”
The preliminary evidence from the study in which I participated shows synchrony between interacting brains and, more intriguingly, that correlations in some brain regions are greater between people while they are telling a joint story than during the independent stories, particularly in the parietal cortex. “That area is active for memory and narrative construction,” Wheatley says. “It seems to fit.”
But the group is also asking whether the content of the stories changes levels of alignment and whether each pair’s relative enjoyment of the process is linked to a greater or lesser degree of synchrony. Like Sid and me, most people reported preferring the joint storytelling exercise to the individual tales, but that wasn’t true for everyone. Are synchronized brains more creative? Or do they just have more fun? The answers will have to wait for further analysis.
One of the challenges of this study is making sense of the mountain of data it generates. Like early astronomers mapping constellations in a star-filled sky for the first time, the scientists have to find order in seeming chaos by making sense of it mathematically. Measuring synchrony is relatively straightforward, Wheatley says, because “we know how to do that math.” The researchers calculate linear correlations between subjects to determine the degree to which parts of their brains respond in the same way over time—are they in lockstep? Does their activity ebb and flow together?
The hyperscanning study is only one way Wheatley is approaching synchrony. In a forthcoming study, available as a preprint, she and Beau Sievers, who is currently working as both a research associate at Harvard and a postdoc at Stanford University, show the power of conversation to synchronize brain patterns. Forty-nine participants watched unfamiliar silent movie clips, then split into small groups of about four people to discuss the clips. Each group was asked to reach a consensus on what the movies were about. After the conversations, the groups watched the clips again, as well as new video from the same movies. After further discussion that reached consensus, patterns of brain processing aligned across participants as they watched the second round of videos. Members of a conversational group had the same brain activity at the same time in brain areas handling vision, memory and language comprehension. The people who listened and worked hardest to seek consensus—and not those who talked most—were the ones whose brains synchronized with others first and who drove synchrony in the larger group. “By talking together and coming to consensus as a group,” Sievers said in a video describing the study, “participants aligned their brains.”
Taken together, these findings are an intriguing way of understanding how our brains facilitate the social interaction that is so critical to human life. Without synchrony and the deeper forms of connection that lie beyond it, we may be at greater risk for mental instability and poor physical health. With synchrony and other levels of neural interaction, humans teach and learn, forge friendships and romances, and cooperate and converse. We are driven to connect, and synchrony is one way our brains help us do it.
Cooperating and conversing are what Sid and I were doing in our separate scanners as we created a story together. More impressive than our effort, though, was that of the pair who came before us. Caitlyn Lee, a graduate student in Wheatley’s lab, was working with Lorie Loeb, a computer science professor at Dartmouth. They set their story not in a park, like ours, but in an unfamiliar landscape. During one of her turns, Lee said, “The trees [the children] were climbing on looked really weird; the ground was starting to rise.” Then her turn cut off, and Loeb picked the story up, saying, “It felt like the creature took a breath.” It was exactly what Lee had been thinking: that the children were walking on the alien itself. “It really felt like we were on the same page,” Lee says.
As we listened to Lee’s retelling, Wheatley turned to me. “At some level,” she said, “I think it has to be the synchrony.”














